Heterogeneous chemistry
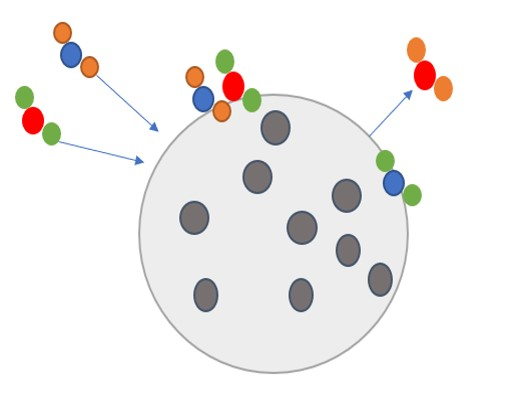
Heterogeneous chemistry describes reactions between compounds in different aggregation phases. Heterogeneous chemical reactions take place e.g. between gases and solid ENMs. Some reactions take place only under the influence of radiation, and reaction products may be released in gas phase or be absorbed to aerosols.
|
|
Occurs in |
|
|
Fate descriptors |
Algorithms |
|
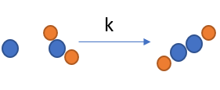
Reaction rate |
|
|
Read more |
Read also |
|
Consult the NanoFASE Library to see abstracts of these deliverable reports: NanoFASE Report D6.2, Open Workshop on airborne ENM: Measurements, Implication and Modelling NanoFASE Report D6.5, Report on model development and evaluation of model quality for airborne ENM |
Ndour et al., GRL 35, L05812, 2008 |
Contact

Astrid Manders
TNO, Netherlands